|
 |
|||||||||||||
|
|||||||||||||
|
|||||||||||||
|
The worlds first horizontal multi-beam klystron has started its site acceptance test at DESY. Built by the Japanese company Toshiba, it is the first of three prototypes from different companies to arrive for the test that will determine whether the new klystron design works. The 10-megawatt horizontal klystron was developed for the European X-ray Free Electron Laser (XFEL) and is also part of the reference design for the ILC. Without klystrons, the particle bunches in the accelerator would not get up to the energies they need to reach in order to collide in the middle at 500 GeV because a crucial driving force - millions of watts of radiofrequency power at 1.3 gigahertz (GHz) waiting to pull the beams along within the superconducting cavities - would not exist. The robotic-looking two-metre tubes are mini particle accelerators themselves. An electron gun shoots pulsed electron beams into its vacuum tube (if you have ever stood next to a klystron: the continuous tic-tic-tic-tic sound it produces is the sound of the pulse transformer). These pass through a low-level radiofrequency signal, which distributes the electron beam into bunches. The bunched electron beam finally generates a high-level radiofrequency signal in an output waveguide. This rf power is transmitted into the accelerator's cavities, waiting to boost the beams on their way to the interaction region. Traditional klystrons have been around in different configurations since the 1940s, and almost all use single round electron beams to amplify the radiofrequency. Multibeam klystrons are rare. The few that exist were mainly constructed in the former Soviet Union. In the 1990s the idea of multibeam klystrons was taken up again for the planned TESLA linear accelerator - that would have used the same SCRF technology designed for the ILC today, and is thus also the technology for the European XFEL. Multibeam klystrons have advantages over single-beam klystrons. They need lower voltage and have at higher efficiency of converting klystron electron beam power to rf power. The reason why they haven't been around for longer is that they pose completely new challenges. “Focussing the beam within the tube is much more complicated,” says Stefan Choroba, klystron expert at DESY. “But since we got the first multibeam prototype in 2000, tested it and analysed its problems the technology has moved along quite a bit.” Multibeam klystrons are now in operation at FLASH and PITZ at DESY. Applications in other fields than in accelerator physics are advancing too, for example in radar technology. No rest for the high-level rf people after tackling the multibeam challenge though. Along with its transformer a klystron measures more than three metres - too high to fit into either the XFEL or the ILC tunnel. The solution? They tilted it. In the XFEL tunnel, where 27 klystrons will tic away when it starts up in three years or so, they sit under the linac itself.
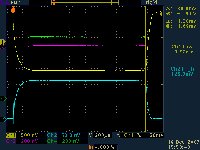
Only it isn't that easy - simply tilting a standard multibeam klystron would be like trying to make baubles on a Christmas tree hang sideways. They weigh three tons, have a cooling system, a magnet and their own cavities, so the design had to be changed quite drastically to get them horizontal. “The magnet and the klystron now form a unit and sit on a single pedestal, rather than the klystron being inserted into the solenoid from the top,” explains Choroba. “The klystron is flanged to the pulse transformer, and the lead shielding has already been integrated into the klystron itself now.” DESY has ordered horizontal multibeam klystron prototypes from three different companies to test them extensively before placing an order for 27 of them in one or two years' time. “We are looking for effects that the manufactures might not see, for example instabilities that only our test setup can detect,” says Choroba. All three prototypes will undergo thorough testing and diagnosis. The first one, after its first week of testing, has already shown an efficiency of 67 percent. Experts from Toshiba, KEK and DESY started to test the tube in December. The scope picture shows measured waveforms of voltage, current and RF power in the two output waveguides. The tests continue in the beginning of the new year in order to analyse all observed data in further detail and to fine tune the klystron. -- Barbara Warmbein |
|||||||||||||
| © International Linear Collider |